
Author: Gilbert H. Jackson, III, BS Chem, JD Law
The author decided to have a sample of amalgam tested after a gold alloy bridge that had been in vivo for 9+ years came loose and fell out of his mouth. An examination of the bottom of the gold alloy surface of the bridge revealed a layer of dark corrosion. An examination of the top of the abutment tooth revealed that it was covered in amalgam. Drawing from his chemistry background the author suspected that a galvanic current between the gold alloy and the amalgam was responsible for the corrosion present on the bottom surface of the gold alloy bridge. A research of the scientific literature revealed conflicting opinions as to whether any mercury leached from amalgam and if it did leach whether there was sufficient release of mercury to have any deleterious health effects. The author researched the methodology for testing the metal content of alloys and determined that energy dispersive spectroscopy coupled with x-ray diffraction analysis was the best method to test the amalgam for its metal content. A small piece of amalgam had come loose from the abutment tooth underneath the gold bridge and this sample measuring 3.00 mm by 1.5 mm and weighing 0.0204 grams was sent to Rice University's geology department for analysis.
A sample of amalgam weighing 0.0204 grams with dimensions of 3.0 mm long by 1.5 mm wide and ranging between 200 micrometers to 500 micrometers in thickness was subjected to qualitative analysis with a PGT energy dispersive spectrometer (EDS) and quantitative (WDS) analysis with a Sun based Cameca SX50 electron microprobe at Rice University's Department of Geology and Geophysics. The testing was performed by Milton L. Pierson, M. Sci, Geology, Microprobe Technician (email: miltonp@ruf.rice.edu) whose collaboration with the author was invaluable in the preparation of the sample and the design of the testing methods.
Rice University's electron microprobe is a Sun computer based Cameca SX50 electron microprobe equipped with a PGT energy dispersive spectrometer (EDS). All the instrument controls (analytical calibration and calculation, digital imaging, and image processing) are done interactively from the console of a Sun Ultra Spark 1 computer with Solaris 2.5.1 operating system and Cameca SXRayN50 software. The computer calculates results for quantitative WDS analyses using the PAP method. All results including digitized images can be transmitted by FTP to the investigator's own computer over the Internet.
The sample can be imaged at magnifications from 63X to 100,000X using techniques similar to scanning electron microscopy while simultaneously viewing the sample by transmitted or reflected light microscopy (400X). The images are collected, processed and stored digitally. Imaging modes include:
1. Secondary electron emission (topography, morphology)
2. Back scattered electron emission (atomic number contrast)
3. X-ray emission (element distribution)
4. Cathodoluminescence (trace element distribution)
5. Absorbed current (electrical conductivity distribution)
Four tests were run on the sample. The bottom of the sample (next to the tooth), the top of the sample (next to the gold alloy bridge), a cross section line analysis and a cross section grid analysis of the sample were tested.
The sample was mounted on a piece of conductive tape in order to provide electrical conductivity. It was then subjected to qualitative analysis and quantitative analysis by a PGT energy dispersive analysis and quantitative analysis with a electron microprobe as set out above. The operating conditions used were a 10 micrometer diameter beam at 15 kV accelerating voltage and 15 nA beam current measured in a mounted faraday cup. The beam current and size were chosen to best quantify the compositional variations within the specimen during data acquisition. The Chemistry present was analyzed by WDS techniques and EDS patterns were collected on representative areas of the specimen. Two sets of 15 microprobes were taken from representative areas of the bottom of the sample and one set of 5 microprobes was taken from an area high in sulfur. The average of the mercury left in one set of 20 probes was 36.34 percent by weight and the average of the mercury left in a second set of 20 microprobes was 41.83 percent by weight. (The data file and the SE and BSE images are shown below as Figure 1, Figure 2 and Figure 3 respectively). Conventional amalgam starts out with approximately 50 percent by weight of mercury. As will be explained later, when you take into account the amount of mercury and the amount of silver that left, considerably more than 8 to 14 % by weight of the mercury left the sample.
| Electron Microprobe Chemical Analyses | ||||||||||
| Amalgam Sample: weight = 0.0204 gram | ||||||||||
| Results expressed as weight % element | ||||||||||
| Element | O | Al | Si | Cu | Zn | Ag | Sn | Hg | Total | |
| #1 | 3.94 | 0.02 | 0.02 | 3.79 | 0.00 | 32.07 | 12.16 | 51.52 | 103.50 | |
| #2 | 3.56 | 0.02 | 0.01 | 4.44 | 0.00 | 46.95 | 18.43 | 27.37 | 100.79 | |
| #3 | 7.68 | 0.04 | 0.01 | 5.98 | 0.53 | 44.62 | 22.15 | 22.45 | 103.47 | |
| #4 | 3.90 | 0.03 | 0.02 | 7.38 | 1.26 | 43.78 | 20.56 | 22.02 | 98.94 | |
| #5 | 4.05 | 0.02 | 0.04 | 1.88 | 0.00 | 33.88 | 9.80 | 51.49 | 101.14 | |
| #6 | 3.21 | 0.03 | 0.06 | 3.47 | 0.00 | 33.60 | 8.31 | 52.18 | 100.84 | |
| #7 | 2.50 | 0.06 | 0.06 | 16.12 | 0.06 | 22.14 | 8.63 | 28.50 | 78.05 | |
| #8 | 3.33 | 0.02 | 0.02 | 3.91 | 0.00 | 41.26 | 12.29 | 39.63 | 100.45 | |
| #9 | 4.56 | 0.03 | 0.02 | 2.69 | 0.00 | 28.16 | 9.39 | 50.76 | 95.60 | |
| #10 | 1.27 | 0.01 | 0.01 | 14.44 | 0.13 | 25.31 | 9.66 | 41.48 | 92.31 | |
| #11 | 4.29 | 0.06 | 0.24 | 16.23 | 23.77 | 3.21 | 5.40 | 13.78 | 66.98 | |
| #12 | 3.23 | 0.02 | 0.02 | 8.29 | 0.08 | 27.05 | 9.53 | 40.18 | 88.40 | |
| #13 | 9.70 | 0.03 | 0.05 | 9.00 | 9.28 | 16.05 | 23.36 | 23.60 | 91.06 | |
| #14 | 2.80 | 0.02 | 0.04 | 2.18 | 0.00 | 36.24 | 9.37 | 45.11 | 95.76 | |
| #15 | 5.58 | 0.03 | 0.07 | 9.27 | 0.55 | 20.87 | 10.67 | 35.03 | 82.06 | |
| O | Al | Si | Cu | Zn | Ag | Sn | Hg | Total | ||
| #1 | 2.66 | 0.03 | 0.01 | 1.73 | 0.00 | 40.10 | 9.91 | 47.38 | 101.83 | |
| #2 | 4.45 | 0.03 | 0.04 | 2.08 | 0.00 | 39.81 | 14.34 | 42.17 | 102.91 | |
| #3 | 3.73 | 0.01 | 0.02 | 2.30 | 0.00 | 41.93 | 12.65 | 41.44 | 102.08 | |
| #4 | 4.21 | 0.02 | 0.02 | 2.87 | 0.00 | 33.84 | 11.63 | 48.76 | 101.34 | |
| #5 | 2.39 | 0.02 | 0.02 | 9.48 | 0.13 | 29.31 | 10.99 | 40.08 | 92.42 | |
| #6 | 3.72 | 0.04 | 0.00 | 1.86 | 0.00 | 44.67 | 12.87 | 40.21 | 103.37 | |
| #7 | 4.98 | 0.04 | 0.02 | 4.70 | 0.62 | 27.81 | 13.44 | 47.89 | 99.50 | |
| #8 | 8.26 | 0.04 | 0.01 | 2.74 | 0.00 | 32.36 | 18.48 | 47.08 | 108.98 | |
| #9 | 12.90 | 0.04 | 0.05 | 3.53 | 0.57 | 27.64 | 29.25 | 25.42 | 99.40 | |
| #10 | 3.03 | 0.06 | 0.15 | 1.98 | 0.00 | 33.20 | 6.06 | 51.10 | 95.58 | |
| #11 | 5.65 | 0.03 | 0.06 | 2.12 | 0.00 | 32.73 | 12.68 | 49.48 | 102.75 | |
| #12 | 4.48 | 0.05 | 0.00 | 5.26 | 0.13 | 46.64 | 19.68 | 25.13 | 101.38 | |
| #13 | 3.78 | 0.07 | 0.05 | 6.04 | 0.62 | 43.48 | 16.65 | 28.05 | 98.74 | |
| #14 | 4.39 | 0.02 | 0.02 | 5.71 | 1.14 | 30.19 | 13.66 | 42.00 | 97.13 | |
| #15 | 5.01 | 0.04 | 0.01 | 1.69 | 0.00 | 33.07 | 12.81 | 51.28 | 103.92 | |
| O | S | Al | Si | Cu | Zn | Ag | Sn | Hg | Total | |
| #1 | 3.43 | 14.80 | 0.00 | 0.02 | 17.26 | 4.11 | 10.43 | 10.21 | 24.48 | 84.74 |
| #2 | 0.00 | 20.85 | 0.01 | 0.02 | 30.83 | 4.33 | 7.55 | 8.56 | 15.91 | 88.05 |
| #3 | 0.71 | 20.69 | 3.09 | 0.01 | 27.16 | 5.75 | 5.75 | 8.10 | 19.88 | 91.15 |
| #4 | 0.60 | 16.10 | 0.09 | 0.02 | 20.78 | 5.43 | 13.68 | 18.08 | 23.77 | 98.55 |
| #5 | 4.04 | 17.76 | 0.01 | 0.03 | 16.96 | 14.01 | 9.71 | 5.89 | 23.33 | 91.74 |
|
FIGURE 2 |

|
|
|
|
FIGURE 3 |

|
|
|
The top of the sample was prepared and mounted in the same manner as the bottom was for Test 1. For the microprobe of the top of the sample an area of the unpolished surface was analyzed by setting up a 100�m by 100�m grid and analyzing 100 evenly spaced points within this grid. The chemistry present was carefully analyzed by WDS techniques on the electron microprobe. Analysis depth is on the order of 1 to 2 �m. Data totals from the unpolished surface were less than 100% due to scattering of the x-ray signal by the irregular surface and the porous nature of this surface. However, an SE image shows the topography and a BSE image shows the average atomic numbers of the metals in the sample and indicate that a high percentage by weight of the mercury has left the surface of the amalgam. These images are not shown in this report.
Test 3-Line analysis of the cross section.
Next, grinding and polishing one side of the piece exposed a cross sectional surface of the amalgam. The grinding and polishing were done using a wet cold method with fine grit wet sandpaper so that the mercury would not be released by heating up the sample during sample preparation. The amalgam was mounted with this polished side up on the carbon tape. Two more sets of data were collected. The first was a line of 100 data points spaced at approximately 2�m intervals beginning on one edge of the amalgam and extending to the opposite edge (Figure 6 below) with a SE, (Figure 4 below) BSE (Figure 5 below)and an x-ray distribution map (Figure 7 below) for the elements that were measured. The x-ray distribution maps clearly show that the areas inside the circles were low in mercury, high in copper, high in tin and high in silver while the matrix outside the circles were high in mercury and low in copper, tin and silver. The average percent by weight of the mercury left in the sample was 28.47%.
| FIGURE 4 |

|
| Figure 4 Scattered Electron (SE)Image of the Cross-section Line Analysis. The line indicates the points from which the 100 microprobes were taken with #1 taken from the top left and point # 100 taken from the bottom right of the line. |
| FIGURE 5 |

|
| Figure 5 Background Scattered Electron (BSE) Image of the Cross-section Line Analysis. Again, the line indicates the points from which the 100 microprobe measurements were taken. |
| FIGURE 6 |

|
| Figure 6 Distribution of Metals in Microprobe Points 1-100 of the Cross-section Line Analysis. |
| FIGURE 7 |

|
| Figure 7 SE, BSE and x-ray diffraction images of the elements S, Cu, O, Sn, C, Si, Zn, Ag and Hg for the Cross-section Line Analysis. For the x-ray diffraction images, the darker areas correspond to areas of relative lower element content and the lighter areas correspond to areas of relative high element content. These images clearly show spheres of low Hg concentration and high Ag, Sn and Cu surrounded by a matrix high in Hg and low in Ag, Sn and Cu. |
Test 4-grid analysis of the cross section.
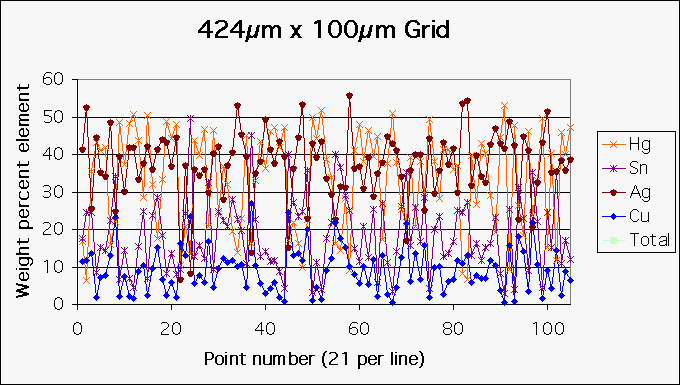
To get a representative measurement of the elements present in the sample a microscopic grid was set up in the thickest part of the sample (approximately 400 micrometers in thickness). This grid measured 424�m by 100�m and consisted of five lines beginning on one edge of the amalgam and extending to the opposite edge spaced 25�m apart. Each line consisted of 21 data points collected at 21�m intervals for a total of 105 data points. The SE image shown in Figure 8 depicts the topography and shows the points at which the microprobe measurements were taken.
| FIGURE 8 |

|
| Figure 8 Scattered Electron (SE) Image of the Cross-section grid analysis. |
The SE image was set up first and the points from which the microprobes were taken were superimposed on the BSE image. The BSE image, shown below in Figure 6, indicates the points at which the microprobe measurements were taken. The measurements for Point 1 was taken from the top, left portion of the top row and the measurements were taken in sequence from left to right until the measurement for point 21 was taken at the far right of the top row. The measurement for Point 22 was taken from the first point in the left side of the second row and proceeded in sequence until 105 measurements were taken over the five rows shown below.
| FIGURE 9 |

|
| Figure 9 Background Scattered Electron (BSE) Image of the Last Grid Analysis. (The dark areas indicate areas with concentration of elements with low mean atomic numbers and the light areas indicate areas with concentrations of high mean atomic numbers. Since mercury ( At. # 80) has the highest atomic number of the metals present in the amalgam, the dark spherical areas indicate areas of low mercury content and the lighter colored matrix outside the spheres indicates areas of high mercury content). |
The graph depicted below as Figure 10 shows the data for the 105 microprobe measurements.
| FIGURE 10 |
|
| Figure 10 Distribution of Metals in Microprobe Points 1-105 of the Cross-section Grid Analysis. |
The table below gives the same information as the graph depicted in Figure 10 above, with the addition of the trace elements O, C, S, Si, Zn and Al.
.
| Amalgam, Polished | |||||||||||||
| Cameca Quantitative Analysis | |||||||||||||
| Label : Amalgam | |||||||||||||
| Sat Sep. 16 0:21:07 2000 | |||||||||||||
| Normal Analysis | |||||||||||||
| Weight Percents | |||||||||||||
| Point # | O | C | S | Hg | Sn | Ag | Zn | Cu | Si | Al | Total | Line # | Point # from left |
| #1 | 0.00 | 1.25 | 0.00 | 16.87 | 17.67 | 41.32 | 0.00 | 11.42 | 0.01 | 0.19 | 88.72 | 1 | 1 |
| #2 | 0.00 | 1.40 | 0.00 | 6.44 | 24.56 | 52.63 | 0.00 | 11.67 | 0.01 | 0.08 | 96.78 | 1 | 2 |
| #3 | 0.00 | 1.98 | 0.00 | 35.48 | 24.41 | 25.67 | 0.00 | 13.61 | 0.02 | 0.04 | 101.21 | 1 | 3 |
| #4 | 0.34 | 1.94 | 0.00 | 43.29 | 8.46 | 44.44 | 0.00 | 1.80 | 0.02 | 0.07 | 100.36 | 1 | 4 |
| #5 | 0.00 | 2.20 | 0.00 | 41.06 | 15.19 | 35.26 | 0.00 | 7.21 | 0.02 | 0.13 | 101.07 | 1 | 5 |
| #6 | 0.00 | 2.16 | 0.00 | 41.97 | 14.44 | 34.19 | 0.00 | 7.86 | 0.02 | 0.07 | 100.70 | 1 | 6 |
| #7 | 0.00 | 1.69 | 0.00 | 15.87 | 22.37 | 48.48 | 0.00 | 13.16 | 0.01 | 0.09 | 101.67 | 1 | 7 |
| #8 | 0.00 | 2.15 | 0.00 | 22.04 | 34.27 | 24.82 | 0.00 | 23.22 | 0.05 | 0.03 | 106.57 | 1 | 8 |
| #9 | 0.36 | 2.00 | 0.00 | 48.48 | 6.74 | 39.53 | 0.00 | 2.24 | 0.02 | 0.07 | 99.44 | 1 | 9 |
| #10 | 6.70 | 4.23 | 0.00 | 37.41 | 14.36 | 30.13 | 0.00 | 7.38 | 0.05 | 3.83 | 104.09 | 1 | 10 |
| #11 | 1.34 | 4.54 | 0.00 | 48.14 | 6.82 | 41.79 | 0.00 | 2.18 | 0.04 | 0.19 | 105.06 | 1 | 11 |
| #12 | 0.74 | 2.66 | 0.00 | 50.60 | 4.59 | 41.80 | 0.00 | 1.54 | 0.03 | 0.08 | 102.03 | 1 | 12 |
| #13 | 0.00 | 1.60 | 0.00 | 43.73 | 15.51 | 33.26 | 0.00 | 8.84 | 0.00 | 0.24 | 103.18 | 1 | 13 |
| #14 | 0.06 | 1.90 | 0.00 | 28.61 | 24.82 | 37.56 | 0.00 | 10.33 | 0.03 | 0.06 | 103.37 | 1 | 14 |
| #15 | 0.76 | 2.16 | 0.00 | 50.32 | 6.83 | 42.19 | 0.00 | 2.28 | 0.03 | 0.12 | 104.69 | 1 | 15 |
| #16 | 0.01 | 0.92 | 0.00 | 31.92 | 23.62 | 36.06 | 0.00 | 9.51 | 0.03 | 0.07 | 102.13 | 1 | 16 |
| #17 | 0.00 | 1.29 | 0.00 | 17.53 | 28.50 | 41.29 | 0.00 | 15.31 | 0.02 | 0.09 | 104.03 | 1 | 17 |
| #18 | 0.19 | 1.61 | 0.00 | 33.33 | 18.46 | 43.90 | 0.00 | 6.57 | 0.02 | 0.08 | 104.15 | 1 | 18 |
| #19 | 0.35 | 1.95 | 0.00 | 48.75 | 8.14 | 43.21 | 0.00 | 2.31 | 0.01 | 0.07 | 104.79 | 1 | 19 |
| #20 | 0.12 | 1.21 | 0.00 | 44.21 | 13.52 | 36.87 | 0.00 | 5.91 | 0.02 | 0.07 | 101.93 | 1 | 20 |
| #21 | 0.97 | 2.04 | 0.00 | 47.93 | 8.03 | 44.43 | 0.00 | 1.93 | 0.03 | 0.21 | 105.58 | 1 | 21 |
| #22 | 14.08 | 9.12 | 12.04 | 10.27 | 9.37 | 6.54 | 6.23 | 16.28 | 0.02 | 4.71 | 88.68 | 2 | 1 |
| #23 | 0.00 | 1.45 | 0.00 | 20.68 | 24.39 | 37.05 | 0.00 | 12.94 | 0.01 | 0.06 | 96.59 | 2 | 2 |
| #24 | 0.00 | 0.98 | 0.00 | 13.21 | 49.57 | 8.30 | 0.00 | 23.56 | 0.03 | 0.03 | 95.68 | 2 | 3 |
| #25 | 0.23 | 2.16 | 0.00 | 43.80 | 12.88 | 36.07 | 0.00 | 5.57 | 0.05 | 0.06 | 100.80 | 2 | 4 |
| #26 | 0.00 | 1.22 | 0.00 | 39.71 | 15.78 | 34.30 | 0.00 | 7.64 | 0.02 | 0.14 | 98.80 | 2 | 5 |
| #27 | 0.00 | 1.57 | 0.00 | 46.74 | 12.19 | 35.99 | 0.00 | 5.75 | 0.02 | 0.05 | 102.32 | 2 | 6 |
| #28 | 0.00 | 1.44 | 0.00 | 20.72 | 32.47 | 29.99 | 0.00 | 16.89 | 0.02 | 0.06 | 101.59 | 2 | 7 |
| #29 | 0.16 | 1.88 | 0.00 | 46.43 | 9.45 | 40.17 | 0.00 | 4.46 | 0.02 | 0.09 | 102.67 | 2 | 8 |
| #30 | 0.06 | 1.32 | 0.00 | 24.39 | 24.90 | 42.13 | 0.00 | 9.61 | 0.03 | 0.15 | 102.58 | 2 | 9 |
| #31 | 0.00 | 1.15 | 0.00 | 36.71 | 22.29 | 28.03 | 0.00 | 12.23 | 0.03 | 0.05 | 100.49 | 2 | 10 |
| #32 | 0.00 | 1.57 | 0.00 | 32.09 | 20.91 | 37.08 | 0.00 | 11.25 | 0.01 | 0.06 | 102.98 | 2 | 11 |
| #33 | 0.00 | 0.74 | 0.00 | 19.43 | 28.04 | 40.49 | 0.00 | 11.64 | 0.01 | 0.06 | 100.41 | 2 | 12 |
| #34 | 0.00 | 1.65 | 0.00 | 13.92 | 22.62 | 53.16 | 0.00 | 10.17 | 0.03 | 0.10 | 101.66 | 2 | 13 |
| #35 | 0.66 | 2.02 | 0.00 | 23.04 | 19.87 | 45.31 | 0.00 | 10.60 | 0.01 | 0.21 | 101.72 | 2 | 14 |
| #36 | 0.55 | 2.32 | 0.00 | 45.02 | 11.06 | 39.39 | 0.00 | 4.41 | 0.02 | 0.10 | 102.88 | 2 | 15 |
| #37 | 0.00 | 1.51 | 0.00 | 16.26 | 45.03 | 13.96 | 0.00 | 26.91 | 0.04 | 0.02 | 103.72 | 2 | 16 |
| #38 | 0.29 | 2.28 | 0.00 | 32.95 | 22.67 | 34.88 | 0.00 | 10.41 | 0.02 | 0.09 | 103.59 | 2 | 17 |
| #39 | 4.46 | 2.87 | 0.00 | 43.44 | 12.90 | 38.13 | 0.00 | 5.49 | 0.05 | 1.46 | 108.81 | 2 | 18 |
| #40 | 0.54 | 1.22 | 0.00 | 36.28 | 14.12 | 49.26 | 0.00 | 2.97 | 0.01 | 0.08 | 104.47 | 2 | 19 |
| #41 | 0.02 | 1.67 | 0.00 | 46.15 | 11.57 | 41.31 | 0.00 | 4.24 | 0.00 | 0.06 | 105.01 | 2 | 20 |
| #42 | 0.14 | 1.45 | 0.00 | 47.25 | 11.67 | 37.55 | 0.00 | 5.96 | 0.04 | 0.06 | 104.11 | 2 | 21 |
| #43 | 1.07 | 1.64 | 0.00 | 41.91 | 8.78 | 43.38 | 0.00 | 1.84 | 0.01 | 0.04 | 98.67 | 3 | 1 |
| #44 | 0.37 | 1.96 | 0.00 | 47.27 | 4.35 | 39.53 | 0.00 | 0.67 | 0.01 | 0.06 | 94.23 | 3 | 2 |
| #45 | 0.00 | 1.05 | 0.00 | 17.03 | 40.03 | 15.20 | 0.00 | 24.63 | 0.04 | 0.07 | 98.05 | 3 | 3 |
| #46 | 0.00 | 1.42 | 0.00 | 21.87 | 27.12 | 36.39 | 0.00 | 13.14 | 0.01 | 0.07 | 100.02 | 3 | 4 |
| #47 | 0.00 | 1.35 | 0.00 | 16.24 | 23.56 | 44.62 | 0.00 | 13.51 | 0.01 | 0.08 | 99.37 | 3 | 5 |
| #48 | 0.00 | 1.50 | 0.00 | 9.87 | 24.73 | 53.31 | 0.00 | 11.76 | 0.00 | 0.09 | 101.25 | 3 | 6 |
| #49 | 0.00 | 0.75 | 0.00 | 22.74 | 35.79 | 23.02 | 0.00 | 20.09 | 0.01 | 0.04 | 102.45 | 3 | 7 |
| #50 | 0.22 | 1.71 | 0.00 | 49.74 | 3.31 | 42.84 | 0.00 | 1.11 | 0.02 | 0.06 | 99.02 | 3 | 8 |
| #51 | 0.41 | 2.02 | 0.00 | 44.32 | 11.14 | 39.19 | 0.00 | 4.44 | 0.00 | 0.07 | 101.59 | 3 | 9 |
| #52 | 0.40 | 2.13 | 0.00 | 51.69 | 3.99 | 43.45 | 0.00 | 1.26 | 0.00 | 0.08 | 103.01 | 3 | 10 |
| #53 | 1.11 | 4.10 | 0.00 | 38.74 | 17.71 | 33.50 | 0.00 | 9.08 | 0.04 | 0.37 | 104.65 | 3 | 11 |
| #54 | 0.00 | 1.03 | 0.00 | 33.96 | 21.91 | 29.24 | 0.00 | 12.23 | 0.04 | 0.06 | 98.48 | 3 | 12 |
| #55 | 0.00 | 0.86 | 0.00 | 16.22 | 40.01 | 22.80 | 0.00 | 21.94 | 0.04 | 0.08 | 101.95 | 3 | 13 |
| #56 | 0.00 | 1.46 | 0.00 | 14.48 | 36.56 | 31.35 | 0.00 | 17.56 | 0.02 | 0.04 | 101.46 | 3 | 14 |
| #57 | 0.00 | 0.89 | 0.00 | 28.25 | 29.19 | 31.29 | 0.00 | 15.08 | 0.01 | 0.07 | 104.77 | 3 | 15 |
| #58 | 0.00 | 1.46 | 0.00 | 9.19 | 24.96 | 55.67 | 0.00 | 10.03 | 0.01 | 0.10 | 101.40 | 3 | 16 |
| #59 | 0.21 | 2.94 | 0.00 | 41.08 | 15.00 | 36.15 | 0.00 | 7.91 | 0.04 | 0.06 | 103.40 | 3 | 17 |
| #60 | 1.01 | 2.87 | 0.00 | 48.13 | 11.39 | 36.68 | 0.00 | 5.68 | 0.04 | 0.09 | 105.88 | 3 | 18 |
| #61 | 7.34 | 9.03 | 0.00 | 38.22 | 20.82 | 30.89 | 0.00 | 10.00 | 0.40 | 0.83 | 117.53 | 3 | 19 |
| #62 | 0.11 | 1.76 | 0.00 | 46.50 | 12.42 | 39.17 | 0.00 | 5.31 | 0.02 | 0.07 | 105.37 | 3 | 20 |
| #63 | 0.88 | 1.92 | 0.00 | 39.47 | 25.24 | 28.76 | 0.00 | 12.02 | 0.09 | 0.14 | 108.51 | 3 | 21 |
| #64 | 1.60 | 1.66 | 0.00 | 44.76 | 5.80 | 35.04 | 0.00 | 2.07 | 0.03 | 0.11 | 91.06 | 4 | 1 |
| #65 | 0.00 | 0.83 | 0.00 | 17.42 | 27.06 | 37.81 | 0.00 | 13.07 | 0.03 | 0.06 | 96.28 | 4 | 2 |
| #66 | 0.15 | 1.47 | 0.00 | 37.58 | 11.19 | 44.88 | 0.00 | 2.63 | 0.02 | 0.08 | 97.98 | 4 | 3 |
| #67 | 0.30 | 0.59 | 0.00 | 51.04 | 3.59 | 43.00 | 0.00 | 0.60 | 0.01 | 0.05 | 99.19 | 4 | 4 |
| #68 | 0.00 | 0.92 | 0.00 | 37.76 | 14.26 | 41.20 | 0.00 | 4.55 | 0.02 | 0.06 | 98.75 | 4 | 5 |
| #69 | 0.00 | 1.30 | 0.00 | 25.37 | 26.06 | 34.25 | 0.00 | 12.58 | 0.01 | 0.06 | 99.63 | 4 | 6 |
| #70 | 0.00 | 1.47 | 0.00 | 21.83 | 35.27 | 17.18 | 0.00 | 21.72 | 0.04 | 0.04 | 97.56 | 4 | 7 |
| #71 | 2.02 | 2.28 | 0.00 | 39.17 | 15.75 | 35.63 | 0.00 | 6.12 | 0.03 | 0.86 | 101.86 | 4 | 8 |
| #72 | 0.00 | 2.06 | 0.00 | 20.86 | 25.95 | 40.03 | 0.00 | 13.47 | 0.03 | 0.08 | 102.48 | 4 | 9 |
| #73 | 0.00 | 1.40 | 0.00 | 38.05 | 16.91 | 39.92 | 0.00 | 6.72 | 0.02 | 0.08 | 103.09 | 4 | 10 |
| #74 | 0.00 | 1.23 | 0.00 | 29.66 | 28.47 | 25.11 | 0.00 | 15.84 | 0.02 | 0.31 | 100.64 | 4 | 11 |
| #75 | 0.50 | 1.49 | 0.00 | 49.45 | 4.33 | 44.23 | 0.00 | 1.82 | 0.02 | 0.09 | 101.92 | 4 | 12 |
| #76 | 0.00 | 1.47 | 0.00 | 38.08 | 19.93 | 29.60 | 0.00 | 9.75 | 0.03 | 0.08 | 98.94 | 4 | 13 |
| #77 | 1.66 | 3.37 | 0.00 | 28.24 | 23.56 | 35.66 | 0.00 | 10.16 | 0.05 | 0.68 | 103.38 | 4 | 14 |
| #78 | 0.30 | 2.56 | 0.00 | 41.28 | 12.87 | 43.30 | 0.00 | 2.78 | 0.02 | 0.07 | 103.18 | 4 | 15 |
| #79 | 0.00 | 1.55 | 0.00 | 41.34 | 13.50 | 37.22 | 0.00 | 6.10 | 0.04 | 0.07 | 99.81 | 4 | 16 |
| #80 | 0.75 | 1.78 | 0.00 | 35.89 | 16.86 | 41.62 | 0.00 | 6.60 | 0.03 | 0.13 | 103.67 | 4 | 17 |
| #81 | 0.00 | 1.47 | 0.00 | 37.07 | 25.13 | 29.97 | 0.00 | 11.68 | 0.05 | 0.10 | 105.46 | 4 | 18 |
| #82 | 0.00 | 1.04 | 0.00 | 8.36 | 24.45 | 53.71 | 0.00 | 11.04 | 0.01 | 0.09 | 98.70 | 4 | 19 |
| #83 | 0.00 | 1.40 | 0.00 | 6.61 | 27.07 | 54.44 | 0.00 | 13.10 | 0.02 | 0.09 | 102.73 | 4 | 20 |
| #84 | 2.23 | 3.91 | 0.00 | 39.72 | 12.98 | 31.72 | 0.00 | 5.78 | 0.15 | 0.73 | 97.22 | 4 | 21 |
| #85 | 0.00 | 1.22 | 0.00 | 26.70 | 16.30 | 39.69 | 0.00 | 7.79 | 0.03 | 0.05 | 91.79 | 5 | 1 |
| #86 | 0.00 | 2.19 | 0.00 | 43.01 | 11.69 | 34.15 | 0.00 | 6.98 | 0.02 | 0.07 | 98.11 | 5 | 2 |
| #87 | 0.53 | 1.53 | 0.00 | 40.22 | 15.19 | 32.58 | 0.00 | 7.00 | 0.01 | 0.23 | 97.29 | 5 | 3 |
| #88 | 0.00 | 1.81 | 0.00 | 29.13 | 16.18 | 42.79 | 0.00 | 11.85 | 0.02 | 0.06 | 101.85 | 5 | 4 |
| #89 | 0.00 | 1.74 | 0.00 | 19.18 | 23.16 | 46.94 | 0.00 | 10.36 | 0.01 | 0.08 | 101.46 | 5 | 5 |
| #90 | 0.86 | 1.71 | 0.00 | 44.56 | 8.15 | 43.02 | 0.00 | 3.85 | 0.02 | 0.31 | 102.48 | 5 | 6 |
| #91 | 0.35 | 2.02 | 0.00 | 53.02 | 2.85 | 41.72 | 0.00 | 0.50 | 0.02 | 0.09 | 100.58 | 5 | 7 |
| #92 | 0.00 | 1.85 | 0.00 | 9.32 | 25.43 | 48.90 | 0.00 | 15.78 | 0.02 | 0.09 | 101.37 | 5 | 8 |
| #93 | 0.61 | 2.14 | 0.00 | 47.74 | 3.90 | 42.49 | 0.00 | 0.69 | 0.02 | 0.08 | 97.67 | 5 | 9 |
| #94 | 0.00 | 0.98 | 0.00 | 26.39 | 31.23 | 22.59 | 0.00 | 18.18 | 0.01 | 0.04 | 99.43 | 5 | 10 |
| #95 | 0.00 | 1.39 | 0.00 | 18.49 | 23.02 | 44.91 | 0.00 | 14.21 | 0.01 | 0.11 | 102.15 | 5 | 11 |
| #96 | 0.39 | 1.96 | 0.00 | 46.25 | 9.08 | 40.97 | 0.00 | 3.46 | 0.03 | 0.08 | 102.22 | 5 | 12 |
| #97 | 0.00 | 1.46 | 0.00 | 23.60 | 35.29 | 20.53 | 0.00 | 21.91 | 0.05 | 0.05 | 102.89 | 5 | 13 |
| #98 | 0.00 | 1.48 | 0.00 | 33.14 | 22.20 | 32.43 | 0.00 | 10.74 | 0.03 | 0.06 | 100.08 | 5 | 14 |
| #99 | 0.61 | 2.55 | 0.00 | 49.71 | 4.50 | 43.23 | 0.00 | 1.51 | 0.03 | 0.05 | 102.19 | 5 | 15 |
| #100 | 0.15 | 1.57 | 0.00 | 15.32 | 24.61 | 51.39 | 0.00 | 8.94 | 0.03 | 0.10 | 102.12 | 5 | 16 |
| #101 | 0.44 | 1.44 | 0.00 | 40.30 | 15.12 | 35.33 | 0.00 | 4.19 | 0.02 | 0.47 | 97.30 | 5 | 17 |
| #102 | 0.00 | 0.78 | 0.00 | 11.85 | 35.58 | 35.52 | 0.00 | 14.27 | 0.01 | 0.06 | 98.08 | 5 | 18 |
| #103 | 0.42 | 1.86 | 0.00 | 45.83 | 10.83 | 38.40 | 0.00 | 2.50 | 0.02 | 0.05 | 99.91 | 5 | 19 |
| #104 | 0.20 | 1.52 | 0.00 | 39.93 | 17.09 | 35.73 | 0.00 | 8.78 | 0.03 | 0.06 | 103.34 | 5 | 20 |
| #105 | 0.00 | 0.97 | 0.00 | 47.32 | 11.98 | 38.69 | 0.00 | 6.30 | 0.03 | 0.04 | 105.34 | 5 | 21 |
| Ave | 0.57 | 1.88 | 0.11 | 33.24 | 18.74 | 37.26 | 0.06 | 9.17 | 0.03 | 0.21 | 101.27 | ||
| Figure 11 Table showing the percent by weight of the elements O, C, S, Hg, Sn, Ag, Zn, Cu, Si and Al in the Cross-Sectional Analysis of the Last Grid. Points #1 through #21 correspond to the top row of points depicted in the SE image shown in Figure 8 and the BSE image shown in Figure 9. Points #22 through #42 correspond to the second row from the top as shown in these images. Each following 21 points correspond to the third, fourth and fifth rows from the top respectively. |
The average percent by weight of the metals that were left in the amalgam were 33.24 % of mercury, 18.74% of tin, 37.26% of silver, 9.17% of copper and .06% of zinc. (See bottom row of Figure 11). The percent by weight of the metals that are contained in conventional silver amalgam are 50% mercury, 35% silver, 10% tin and 3 to 5% copper and a trace of zinc. (See bottom row of Figure 11). These percentages correlate well with the percentages by weight shown in points 1, 5, and 6 of the top 15 microprobes of the bottom of the sample shown in Figure 1 above. The percent by weight of mercury at these points was approximately 50% and the totals add up to approximately 100% and the data from these points are reliable and are indicative of the metal content of the silver amalgam in the original mixture before the mercury and silver leached out. This is strong evidence that the original amalgam in the sample was conventional amalgam.
DISCUSSION
The author decided to do a microprobe of the cross section of the amalgam after reading the full article by M. Hanson and J. Pleva, titled "The Dental Amalgam Issue. A review", Experientia, 1991 Jan 15;47(1):9-22, PMID: 1999251 was procured and read by the author. (The importance of reading the full article rather than just the abstract cannot be overemphasized). M. Hanson and J. Pleva performed a scanning electron microscopy with energy dispersive x-ray analysis of 20 used amalgam dental restorations. One of the images (presumably a BSE image) showed a cross-section through an amalgam filling under a gold alloy crown that had been in vivo for 9 years. This image showed that a layer 100 micrometers thick underneath the gold crown had been depleted of its mercury (presumably due to the galvanic current between the gold alloy crown and the amalgam). Hanson and Pleva reported that this resulted in approximately 30 micrograms of mercury being released per day and that selective corrosion within the amalgam (presumably due to galvanic current between the different phases of amalgam in the filling) was estimated to have released another 30 micrograms of mercury per day for a total of 60 micrograms of mercury released per day from the one tooth.
The author had expected the same results for his sample as was reported by Hanson and Pleva, above, because his sample of amalgam had been underneath a gold alloy bridge for nine+ years in vivo. However, although there was a layer of corroded amalgam low in mercury about 5 micrometers thick on the top surface of the amalgam which was next to the gold alloy bridge, there was no layer of amalgam 100 micrometers thick that was depleted in mercury. Instead there were spheres varying between 20 micrometers and 40 micrometer in diameter which were low in mercury, high in tin, high in copper, and high in silver surrounded by a matrix with a mercury content of approximately 40% to 50 % by weight. A possible explanation for the different results will be explained later.
A. Mercury Release due to selective corrosion within the amalgam.
The grid analysis of the cross section revealed that the average percent by weight of the mercury decreased to approximately 33%, the average percent by weight of the tin increased to approximately 19%, almost double the initial 10%, the average percent by weight of the silver stayed about the same, approximately 35%, and the average percent by weight of the copper increased to approximately 9% by weight, almost double its initial 3 to 5%. Since mercury is the heaviest of the metals (molecular weight of 200.59), as it leaves you would expect the percentages by weight of the metals that stayed behind to increase. Both the percentages by weight of the tin and the copper increased but the silver stayed about the same. This indicates that silver and mercury both leach out. My research indicates that the amount of silver that is found in the tissues is a good indicator of the amount of mercury that is in the tissues in people with silver amalgam fillings and no other apparent source of silver or mercury. The source is "Silver concentrations in human tissues, their dependence on dental amalgam and other factors", J. Trace Elem Med. Biol 1995 Jul; 9(2):82-7, PMID: 8825980 by G. Drasch and others. Hanson and Pleva cited above also indicates that tin does not leach out but stays behind as corrosion products. In order to compute the actual percent by weight amount of mercury and silver that leached out, the weight of the sample before the metals leached out must be computed.
One method that I used to compute the original weight of the sample was to multiply the weight of the sample by the percent of tin left in the sample as follows:
20.4 mg(weight of sample) x .19 ( % of tin left) = 3.876 mg.
Since the original sample contained .10 tin, I then solved for the equation:
.10 x = 3.876 mg where x = the original weight of the sample Solving for x gives 38.76 mg. So the original sample weighed 38.76 mg. To compute the original percentage of copper in the sample you multiply .09 (average percent of copper left) times 20.4 mg(weight of the sample) and divide the result by 38.76 mg to get .047. The original percent by weight of the copper was 4.7%. Doing the same for zinc would give .00016 or .016% as the original percent by weight (a trace). The percentage of mercury by weight is typically 50% and the percentage of silver by weight is typically 35%.
Based on these assumptions and calculations the original percent by weight would be 50% mercury, 35% silver, 10% tin, 4.7% copper and .016% zinc. To calculate the amount of mercury and silver that left the sample you compute as follows:
For mercury: .50 x 38.76 mg. (original weight) = 19.38 mg. (original weight of mercury)
.33 x 20.40 mg. (sample weight) = 6.73 mg. (weight of mercury left in sample)
19.38 mg. - 6.73 mg. = 12.65 mg. of mercury that left the original sample.
12.65 mg. divided by 19.38 mg = .65.
65% of the mercury that was in the original sample leached out.
For silver: .35 x 38.76 mg. (original weight) = 13.56 mg. (original weight of silver)
.37 x 20.40 mg. (sample weight) = 7.55 mg. (weight of silver left in sample)
13.56 mg. - 7.55 mg. = 6.01 mg. of the silver that left the original sample.
6.01 divided by 13.56 mg. = .44
44% of the silver that was in the original sample leached out.
The sample that was used was approximately 3 mm. by 1.5 mm. The surface area of the silver amalgam on the abutment tooth that the sample came from covered approximately 8 times the area of the sample. Assuming that the rest of the amalgam covering the abutment tooth was approximately the same average thickness then one can estimate that 101.2 mg of mercury left the silver amalgam covering the abutment tooth over 9 + years and that 48.08 mg of silver left the silver amalgam over 9 + years.
If you divide these weights by the molecular weight of the respective metals as follows:
.1012 gm. divided by 200.59 gm./mole = .0005045 moles of mercury left over 9 + years.
.04808 gm. divided by 107.86 gm./mole = .0004458 moles of silver left over 9+ years. The mercury and silver left on an approximate 1 to 1 ratio. For every molecule of mercury that left one molecule of silver left. This correlates well with what a materials science professor explained to me which was that the mercury and silver would leave on a 1 to 1 ratio until a phase was left in which the silver and mercury were bound on the atomic level.
To compute the daily release of mercury and silver over approximately 10 years you compute as follows:
For mercury:
101.2 mg. divided by (10 years x 365 days/year) = .0277 mg. = 28 micrograms/day
For silver:
48.08 mg. divided by (10 years x 365 days/year) = .0132 mg. = 13 micrograms/day
28 micrograms of mercury released per day correlates well with the 30 micrograms of mercury per day that Hanson and Pleva estimated to have been released per day due to selective corrosion within the amalgam that was underneath the gold crown.
I will now propose an explanation of the circles of low mercury and high silver, copper, and tin surrounded by the matrix of high mercury. Hanson and Pleva describe the amalgamation reaction in simplified version as:
AG3Sn + Hg --> Ag2Hg3 + Sn6-8Hg or
gamma-phase +Hg --> gamma-1 +gamma-2-phase
The gamma-1 phase can be a number of combinations of Ag3Hg4, Ag4Hg5, Ag2Hg3, Ag11Hg15 and Ag5Hg8. The gamma-2 phase can be Sn7Hg, Sn8Hg, Sn7-8Hg and Sn6Hg and is the phase that is prone to corrosion. The amount of gamma-2 phase can vary between zero and 59%, but 15-20% is common and is dependent on the original composition of the amalgam mixture, the trituration time and insertion technique of the dentist.
"The Dental Amalgam Issue. A review", Experientia, 1991 Jan 15;47(1):9-22, p10.
If you look at point 12 in the Grid Analysis of the Cross Section (Figure 11 above) which is in the high Hg area, you have an approximate ratio by weight of 4 Ag to 5 Hg which when you take into account the molecular weights you would get Ag8Hg5. If you look at the normal reaction product for the gamma-1 phase, Ag2Hg3 you would expect a percent by weight ratio of 1 Ag to 3 Hg and actually all of the other gamma-1 phases have an approximate percent by weight ratio of 1 Ag to 3 Hg. If the high mercury areas surrounding the spherical high Sn, low Hg areas is the gamma-1 phase, then it should consist of Hg that is bound to Ag in a ratio of 3 Hg to 1 Ag by weight with mercury and silver being released on a 1 to 1 ratio as was shown in the calculations above. However, there is a lot more silver in the matrix than would be expected in a gamma 1 phase.
If you look at the gamma-2 phases of Sn6Hg, Sn7Hg and Sn8Hg and take into account the molecular weight of the two metals, you would have a 3.5 Sn to 1 Hg ratio, a 4 Sn to 1 Hg ratio and a 5 Sn to 1 Hg ratio, respectively. Point # 83 of the Grid Analysis of the Cross Section (Figure 11 above) which is in the spherical areas of low Hg and high Sn has a ratio by weight of 4 Sn to 1 Hg and is probably the gamma-2 phase. However, this point also has 54% Ag by weight where you would expect no silver if it were the gamma-2 phase. The ratio of Ag to Hg at point # 83 is 8 Ag to 1 Hg. This is 24 times more silver than is expected in a gamma-1 phase.
A possible explanation is that the gamma -2 phase is evenly distributed throughout the amalgam when the amalgam is first mixed. The areas of the gamma-2 phase and the gamma-1 phase which would have different electrochemical potentials because of the different compositions of metals would set up many small galvanic cells which would release mercury and silver on a 1 to 1 ratio as explained above. As time went by and the galvanic currents continued the area of gamma-2 would increase in size to the spheres of low mercury and high tin that is seen in the BSE images and mercury and silver would continue to be released. Although the percentage of mercury is 50% by weight and the percentage of silver is 35% by weight, the actual percentage by molecules is 34 % mercury to 44% silver. As mercury and silver are released on a 1 to 1 basis as shown by the calculations above, the percent by weight of silver would increase because of the silver that was left behind due to the 10% difference in their percentages by molecule. This explanation is supported by the fact that both point # 12 presumably in the gamma-1 phase and point # 83, presumably in the gamma-2 phase both have higher percentages by weight of silver than would be expected to be present in the two phases.
When you have the different phases in the amalgam, you get a galvanic current set up between the different phases and get corrosion of the amalgam and release of mercury. If the
electrochemical potential difference between the two phases is greater than the electrochemical potential difference between the gold and the amalgam phase that it is in contact with, then the galvanic current would flow between the amalgam phases and the gold/amalgam interface would be protected, no galvanic current would take place there. In any event the low Cu amalgam with approximately 5% Cu which is what we have and which produces the gamma-2 phase is known to corrode and deteriorate.
Hanson and Pleva also state that the corrosion products of silver amalgam consist mainly of hydroxy-chlorides and oxides of tin. The x-ray distribution maps indicate to me that the spherical portions all have relatively high concentration of Ag, Sn and Cu in that order of percent by weight while they have almost no Hg. The areas outside the spherical portions contain by weight
relatively high proportions of Hg and moderately high Ag but almost no Sn or Cu.
.
B. Mercury release from dental amalgam due to galvanic current between different metals in dental restorative materials.
This investigation into mercury loss from amalgam was sparked when a gold alloy bridge came off of its abutment teeth and the author noticed that the bottom of the bridge had corrosion on it where it had been glued to the molar abutment tooth. All of the glue that had been used to glue the bridge in place was missing from the molar abutment tooth and the bridge surface that came into contact with it. There was glue present on the bridge where it had been glued to the front abutment tooth. An examination of the molar abutment tooth revealed that it was covered with amalgam. From his chemistry background the author knew that when dissimilar metals are in contact a galvanic current is set up with a loss of metal at the sacrificial anode. A research of medline revealed 8 articles when the search words (gold AND amalgam AND "galvanic current") were entered. One of these articles reported that a current density of 200 microA/dm2 existed between conventional amalgam and a casting gold allow type III. The article was Galvanic currents between dental alloys in vitro" Scand J Dent Res 1985 Oct;93(5):467-73, PMID # 3864220 by k Arvidson and J. Johansson.
After determining the electrochemical potentials of the metals in amalgam from the CRC handbook the daily release of mercury was computed using the 200 microamp/dm2 current density and Faraday's law. The electrochemical potentials that I found are as below in the order
from most reactive (+) to least reactive (-):
Zn & Zn(Hg) > Sn > Cu(Hg) > Cu > Ag > Hg > Au
+.7626 +.1406 -.3511 -.3400 -.799 -.852 -1.42
A current density of 200 microamp/dm2 for a contact surface of 6 mm square results in a current of .72 microamp. For the sake of ease of calculation .72 microamp was rounded off to 1 microamp.
10 years x 365 days x 24 hours x 60 min x 60 sec = 315,360,000 sec
A 1 microamp current constant for10 years would produce 315,360,000 microA sec. which equals 315 amp. sec. or coulombs. Faraday's Number is 96,500 Coulombs and is the current necessary to produce 1 mole of electrons ( Avogadro' number or 6.02 times 10 to the 23rd power) and a Coulomb is an ampere sec. Using Faraday's number and taking account that Zn, Cu and Sn all produce ions with a double positive charge and that they all would result in the release of the Hg that was bound to them, the calculation would be as follows:
315 Coulombs divided by (96,500 Coulombs x 2) = .0016321 moles of Hg
being released. A mole of a material is the molecular weight expressed in grams and contains Avogadro's number of molecules (6.02 times 10 to the 23rd power)
.0016321 moles x 200.59 gms/mole for Hg = .327 gms of mercury
released over 10 years.
To get the daily mercury burden in micrograms I computed as follows:
.327 gms of Hg = 327,000 micrograms which you divide by 10 years and 365
days to get a daily burden of 89.6 micrograms of Hg from one 6mm square contact surface between a gold alloy crown/bridge and an amalgam surface to which it has been glued. If you have four of these then theoretically you could be getting 360 micrograms of mercury per day
from the galvanic reaction. If it is released in the form of mercury vapor, it would be absorbed directly into the blood stream and go to the brain. Since the OSH PEL limit is 50 micrograms per cubic meter for air, 360 micrograms released into the blood stream would certainly cause
problems.
After doing these calculations and acknowledging the different opinions in the literature as to whether silver amalgam was a stable alloy, the author decided to have a 3 mm. by 1.5 mm. sample of the amalgam which had fallen off of the amalgam covered abutment tooth analyzed for mercury and other metals to determine for himself whether any of the mercury in the amalgam was released due to the galvanic current between the gold alloy bridge and the silver amalgam to which it was glued. The results as shown above suggest that there was no appreciable loss of metals due to the galvanic current between the gold alloy and the amalgam but that there was an appreciable loss of mercury (approximately 30 micrograms per day) and an appreciable loss of silver (approximately 13 micrograms per day) due to the corrosion from the galvanic current within the amalgam. There are a couple of possible explanations for the lack of corrosion due to galvanic current between the gold alloy bridge and the amalgam underneath it. One is that the combinations of metals in the gold alloy bridge has an electrochemical potential that does not create a galvanic current between the gold and the amalgam. In "Galvanic currents between dental alloys in vitro" Scand J Dent Res 1985 Oct;93(5):467-73 by K. Arvidson and J. Johansson only one of the gold alloys tested, the type III Sjoding C with a composition of 76.5% gold, 11.5% copper, 8.5% silver, 0.5% platinum and a trace of zinc showed an appreciable current density with conventional amalgam-200 microA/dm2. The composition of the gold alloy bridge is not yet known and may be one of the ones that do not show an appreciable galvanic current with conventional amalgam.
Another explanation is that a competing galvanic current protected the gold alloy amalgam interface. A steady 2 microamp current was measured between the gold alloy bridge glued to the abutment tooth on the lower jaw from which the sample was taken and a gold crown on the other side of the lower jaw. This 2 microamp current would protect the gold amalgam interface which theoretically could produce a 1 microamp current. The author has three other gold alloy bridge/crowns with amalgam couplings and will have the amalgam tested to determine whether there has been any mercury and silver loss due to galvanic current between the gold alloys and the amalgam and due to galvanic current between the different phases within the amalgam.
CONCLUSION
The micro probe analysis, BE, BSE and x-ray distribution maps are compelling evidence that mercury and silver are released from dental amalgam and that dental amalgam is not a stable alloy. 65% of the mercury and 44% of the silver that was in the amalgam when it was first prepared leached out over 9 + years. Approximately 30 micrograms of mercury was calculated to be released per day and 13 micrograms of silver were calculated to be released per day. Over the 9+ year life of the amalgam, 101.2 mg of mercury left the silver amalgam covering the abutment tooth and 48.08 mg of silver left the silver amalgam. No one can seriously argue that 101.2 mg of mercury in the body isn't harmful. Faraday's law of 96,500 coulombs (ampere seconds) can be used to show that a 1 microamp current between a gold alloy crown or bridge and an amalgam filling that are in continuous contact for 10 years will release 90 micrograms of mercury per day. Since aged amalgam has not been tested by the scientific community in the United States, it is suggested that any individual, any interested dentist, chemist, material science experts, and metallurgists save any amalgam that is removed from their teeth and to send it in for analysis and send the results to this web site so that a body of evidence can be gathered. As the author has his amalgam removed, he will have it tested and will add the results to this web site. Milton L. Pierson, M. Sci, Geology, Micro probe Technician (email: miltonp@ruf.rice.edu) is an excellent resource for having the amalgam tested. The analysis of this data by the author is a beginning and is by no means the definitive analysis. Analysis by chemists, dentists, metallurgists, material scientists and individuals is welcome and may be directed to the author at the email address below.
Gilbert H. Jackson, III, BS Chemistry (Phi Beta Kappa, UNC 1969) JD Law (UNC 1982).
Email address: giljack@mindspring.com
Special thanks to Professor Boyd Haley, Chair of the Chemistry
Department at the University of Kentucky for reviewing the data files and images used in this report.
ADDENDUM Anyone interested in learning more about galvanic current between gold alloys and
amalgam and between amalgams of different compositions should read the full text of the 8
articles that can be found on PubMed by using the search words "gold AND amalgam AND
"galvanic current". Two of those have been cited in the text above and are listed below with the
other 6 and are as follows: 1. "The dental amalgam issue. A review", Experientia. 1991 Jan 15;47(1):9-22. Review
by M. Hanson and J. Pleva.
1999251. 2. "Galvanic currents between dental alloys in vitro" Scand J Dent Res 1985
Oct;93(5):467-73 by k Arvidson and J. Johansson.
3864220. 3. "A measurement of galvanic current and electrical potential in extracted human teeth",
J. Dent. Res 65(12):1441-1444, December, 1986 by Y. Momoi and others.
3465773 4. "Galvanic currents between gold and amalgam", Scand. J. Dent. Res. 1980: 88:269-272 by R. I. Holland.
6932094 5. "A galvanic study of different amalgams", Journal of Oral Rehabilitation 1977, Vol. 4,
pp. 23-27 by C. P. Wang Chen and E. H. Greener.
265360 6. "A Histological study of the influence of galvanic current between metal alloys in
connective tissue in the rat", 1973, Proc. Fin. J. Dent. Soc. 69: 1-6, by Tuija Parvinen and
Antti Yli-Urpo.
4731447 7. "Clinical management of galvanic current between gold and amalgam", Gen Dent.
1996 Jan-Feb;44(1):70-3. Review by R. Williamson.
8940574 8. "Intraoral corrosion resulting from coupling dental implants and restorative metallic
systems", Implant Dent. 1992 summer;1(2):107-112 by J.E. Lemons and others.
1288802 LINKS
.
.